 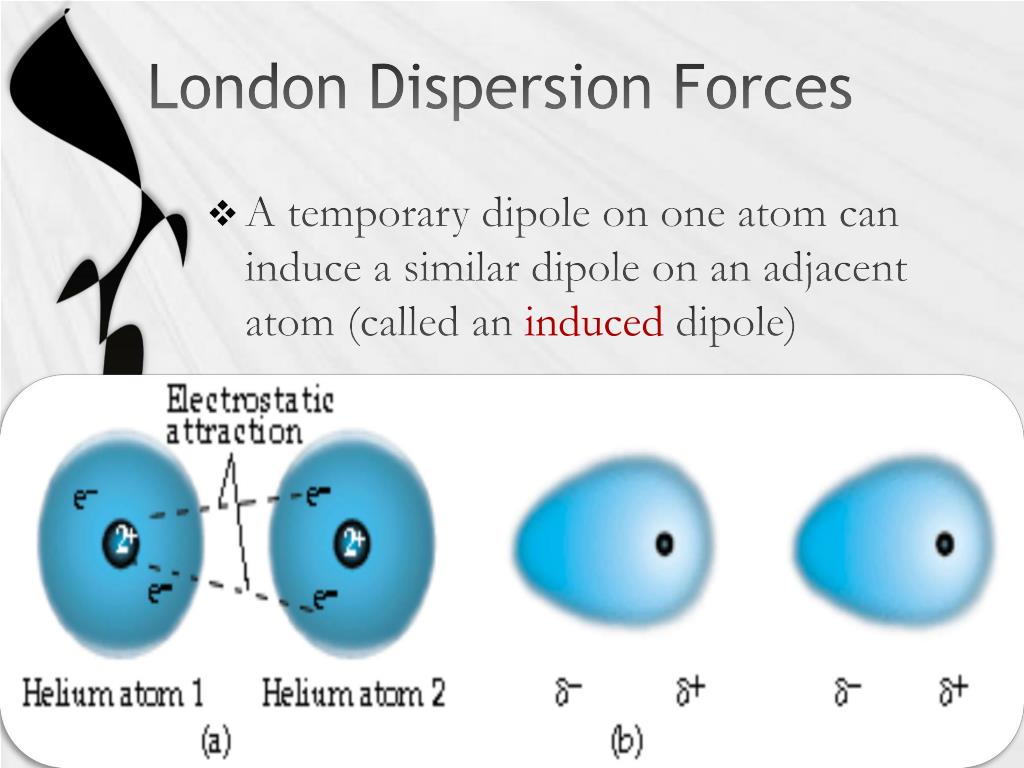
The magnitude of the London dispersion force is frequently described in terms of a single parameter called the Hamaker constant, typically symbolized A : CS1 maint: multiple names: authors list ( link) While the detailed theory requires a quantum-mechanical explanation (see quantum mechanical theory of dispersion forces), the effect is frequently described as the formation of instantaneous dipoles that (when separated by vacuum) attract each other. The net effect is that the fluctuations in electron positions in one atom induce a corresponding redistribution of electrons in other atoms, such that the electron motions become correlated. These fluctuations create instantaneous electric fields which are felt by other nearby atoms and molecules, which in turn adjust the spatial distribution of their own electrons. 
The electron distribution around an atom or molecule undergoes fluctuations in time. They are the weakest intermolecular force. The LDF is named after the German physicist Fritz London. They are part of the van der Waals forces. London dispersion forces ( LDF, also known as dispersion forces, London forces, instantaneous dipole–induced dipole forces, fluctuating induced dipole bonds or loosely as van der Waals forces) are a type of intermolecular force acting between atoms and molecules that are normally electrically symmetric that is, the electrons are symmetrically distributed with respect to the nucleus. The long-range section is due to London dispersion forces. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
